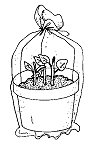Introduction
Propagation by stem cuttings is the most commonly used method to propagate many woody ornamental plants. Stem cuttings of many favorite shrubs are quite easy to root. Typically, stem cuttings of tree species are more difficult to root. However, cuttings from trees such as crape myrtles, some elms, and birches can be rooted.
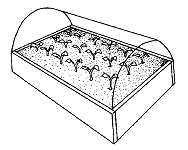
A greenhouse is not necessary for successful propagation by stem cuttings; however, maintaining high humidity around the cutting is critical. If rooting only a few cuttings, you can use a flower pot (Figure 1). Maintain high humidity by covering the pot with a bottomless milk jug or by placing the pot into a clear plastic bag. Cuttings can also be placed in plastic trays covered with clear plastic stretched over a wire frame (Figure 2). Trays must have holes in the bottoms for drainage. The plastic will help keep the humidity high and reduce water loss from the cuttings.
If you need more elaborate facilities, you can construct a small hoop frame and/or use an intermittent mist system. HIL-404, Low Investment Propagation / Winter Protection Structure, and HIL-405, A Simple Intermittent Mist System For Propagation, describe how this can be accomplished. Another publication that may be helpful is AG-426, A Small Backyard Greenhouse for the Home Gardener.
Types of Stem Cuttings
The four main types of stem cuttings are herbaceous, softwood, semi-hardwood, and hardwood. These terms reflect the growth stage of the stock plant, which is one of the most important factors influencing whether or not cuttings will root. Calendar dates are useful only as guidelines. Refer to Table 1 for more information on the best time to root stem cuttings of particular ornamental plants.
| Common Name | Scientific Name | Type of Cutting (SW = softwood, SH = semi-hardwood, HW = hardwood) |
|---|---|---|
| Evergreen Plants | ||
| Abelia | Abelia spp. | SH, HW |
| Arborvitae, American | Thuja occidentalis | SH, HW |
| Arborvitae, Oriental | Platycladus orientalis | SW |
| Azalea (evergreen & semi-evergreen) | Rhododendron spp. | SH |
| Barberry, Mentor | Berberis x mentorensis | SH |
| Barberry, Japanese | Berberis thunbergii | SH, HW |
| Barberry, wintergreen | Berberis julianae | SH |
| Boxwood, littleleaf | Buxus microphylla | SH, HW |
| Boxwood, common | Buxus sempervirens | SH, HW |
| Camelia | Camelia spp. | SW, SH, HW |
| Ceanothus | Ceanothus spp. | SW, SH, HW |
| Cedar | Cedrus spp. | SH, HW |
| Chamaecyparis; False cypress | Chamaecyparis spp. | SH, HW |
| Cotoneaster | Cotoneaster spp. | SW, SH |
| Cryptomeria, Japanese | Cryptomeria japonica | SH |
| Daphne | Daphne spp. | SH |
| Elaeagnus, thorny | Elaeagnus pungens | SH |
| English ivy | Hedera helix | SH, HW |
| Euonymus | Euonymus spp. | SH |
| Fir | Abies spp. | SW, HW |
| Gardenia; Cape jasmine | Gardenia jasminoides | SW, SH |
| Heath | Erica spp. | SW, SH |
| Hemlock | Tsuga spp. | SW, SH, HW |
| Holly, Chinese | Ilex cornuta | SH, HW |
| Holly, Foster's | Ilex x attenuata 'Fosteri' | SH |
| Holly, American | Ilex opaca | SH |
| Holly, Yaupon | Ilex vomitoria | SH, HW |
| Holly, English | Ilex aquifolium | SH |
| Holly, Japanese | Ilex crenata | SH, HW |
| Jasmine | Jasminum spp. | SH |
| Juniper, creeping | Juniperus horizontalis | SH, HW |
| Juniper, Chinese | Juniperus Chinensis | SH, HW |
| Juniper, shore | Juniperus conferta | SH, HW |
| Leyland cypress | x Cupressocyparis leylandii | SH, HW |
| Magnolia | Mahonia spp. | SH |
| Oleander | Nerium oleander | SH |
| Osmanthus, holly | Osmanthus heterophyllus | Sh, HW |
| Photinia | Photinia spp. | SH, HW |
| Pine, Mugo | Pinus mugo | SH |
| Pine, Eastern white | Pinus strobus | HW |
| Pittosporum | Pittosporum spp. | SH |
| Podocarpus | Podocarpus spp. | SH |
| Privet | Ligustrunum spp. | SW, SH, HW |
| Pyracantha; Firethorn | Pyracantha spp. | SH |
| Rhododendron | Rhododendron spp. | SH, HW |
| Spruce | Picea spp. | SW, HW |
| Viburnum | Viburnum spp. | SW, HW |
| Yew | Taxus spp. | SH, HW |
| Common Name | Scientific Name | Type of Cutting (SW = softwood, SH = semi-hardwood, HW = hardwood) |
| Deciduous Trees | ||
| Azalea (deciduous) | Rhododendron spp. | SW |
| Basswood; American linden | Tilia americana | SW |
| Birch | Betula spp. | SW |
| Bittersweet | Celastrus spp. | SW, SH, HW |
| Blueberry | Vaccinium spp. | SW, HW |
| Broom | Cytisus spp. | SW, HW |
| Callery pear | Pyrus calleryana | SH |
| Catalpa | Catalpa spp. | SW |
| Clematis | Clematis spp. | SW, SH |
| Crabapple | Malus app. | SW, SH |
| Crape myrtle | Lagerstroemia indica | SH |
| Cherry, flowering | Prunus spp. | SW, SH |
| Dawn redwood | Metasequoia glyptostroboides | SW, SH |
| Deutzia | Deutzia spp. | SW, HW |
| Dogwood | Cornus spp. | SW, SH |
| Elderberry | Sambucus spp. | SW |
| Elm | Ulmus spp. | SW |
| Euonymus | Euonymus spp. | HW |
| Forsythia | Forsythia spp. | SW, SH, HW |
| Fringe tree | Chioanthus spp. | SW |
| Ginkgo, Maidenhair tree | Ginkgo biloba | SW |
| Goldenrain tree | Koelreuteria spp. | SW |
| Hibiscus, Chinese | Hibiscus rosa-sinensis | SW, SH |
| Honey locust | Gleditsia triacanthos | HW |
| Honeysuckle | Lonicera spp. | SW, HW |
| Hydrangea | Hydrangea spp. | SW, HW |
| Ivy, Boston | Parthenocussus tricuspidata | SW, HW |
| Larch | Larix spp. | SW |
| Lilac | Syringa spp. | SW |
| Maple | Acer spp. | SW, SH |
| Mock orange | Philadelphus spp. | SW, HW |
| Mulberry | Morus spp. | SW |
| Poplar; Aspen; Cottonwood | Populus spp. | SW, HW |
| Poplar, Yellow; Tulip tree; Tulip poplar | Liriodendron tulipfera | SH |
| Quince, flowering | Chaenomeles spp. | SH |
| Redbud | Cercis spp. | SW |
| Rose of Sharon; Shrub-althea | Hibiscus syriacus | SW, HW |
| Rose | Rosa spp. | SW, SH, HW |
| Russian olive | Elaeagnus angustifolia | HW |
| Serviceberry | Amelanchier spp. | SW |
| Smoke tree | Cotinus coggygria | SW |
| Spirea | Spiraea spp. | SW |
| St. Johnswort | Hypericum spp. | SW |
| Sumac | Rhus spp. | SW |
| Sweet gum | Liquidambar styraciflua | SW |
| Trumpet creeper | Campsis spp. | SW, SH, HW |
| Virginia creeper | Parthenocissus quinquefolia | SW, HW |
| Weigela | Weigela spp. | SW, HW |
| Willow | Salix app. | SW, SH, HW |
| Wisteria | Wisteria spp. | SW |
Herbaceous cuttings are made from non-woody, herbaceous plants such as coleus, chrysanthemums, and dahlia. A 3- to 5-inch piece of stem is cut from the parent plant. The leaves on the lower one-third to one-half of the stem are removed. A high percentage of the cuttings root, and they do so quickly.
Softwood cuttings are prepared from soft, succulent, new growth of woody plants, just as it begins to harden (mature). Shoots are suitable for making softwood cuttings when they can be snapped easily when bent and when they still have a gradation of leaf size (oldest leaves are mature while newest leaves are still small). For most woody plants, this stage occurs in May, June, or July. The soft shoots are quite tender, and extra care must be taken to keep them from drying out. The extra effort pays off, because they root quickly.
Semi-hardwood cuttings are usually prepared from partially mature wood of the current season’s growth, just after a flush of growth. This type of cutting normally is made from mid-July to early fall. The wood is reasonably firm and the leaves of mature size. Many broadleaf evergreen shrubs and some conifers are propagated by this method.
Hardwood cuttings are taken from dormant, mature stems in late fall, winter, or early spring. Plants generally are fully dormant with no obvious signs of active growth. The wood is firm and does not bend easily. Hardwood cuttings are used most often for deciduous shrubs but can be used for many evergreens. Examples of plants propagated at the hardwood stage include forsythia, privet, fig, grape, and spirea.
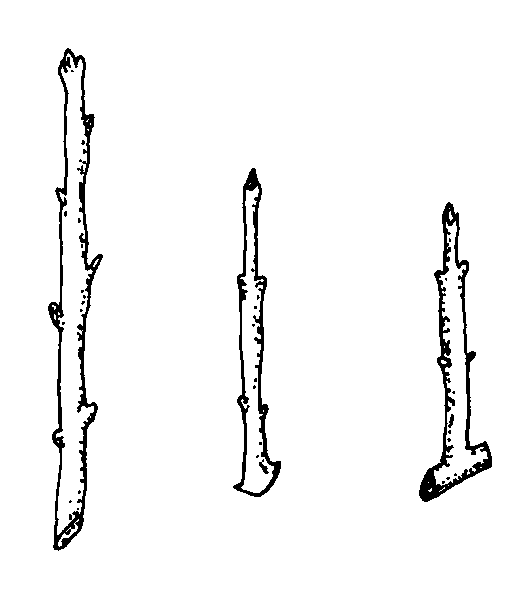
The three types of hardwood cuttings are straight, mallet, and heel (Figure 3). A straight cutting is the most commonly used stem cutting. Mallet and heel cuttings are used for plants that might otherwise be more difficult to root. For the heel cutting, a small section of older wood is included at the base of the cutting. For the mallet cutting, an entire section of older stem wood is included.
Procedures for Rooting Stem Cuttings
Cuttings should generally consist of the current or past season’s growth. Avoid material with flower buds if possible. Remove any flowers and flower buds when preparing cuttings so the cutting’s energy can be used in producing new roots rather than flowers. Take cuttings from healthy, disease-free plants, preferably from the upper part of the plant.
The fertility status of the stock (parent) plant can influence rooting. Avoid taking cuttings from plants that show symptoms of mineral nutrient deficiency. Conversely, plants that have been fertilized heavily, particularly with nitrogen, may not root well. The stock plant should not be under moisture stress. In general, cuttings taken from young plants root in higher percentages than cuttings taken from older, more mature plants. Cuttings from lateral shoots often root better than cuttings from terminal shoots.
Early morning is the best time to take cuttings, because the plant is fully turgid. It is important to keep the cuttings cool and moist until they are stuck. An ice chest or dark plastic bag with wet paper towels may be used to store cuttings. If there will be a delay in sticking cuttings, store them in a plastic bag in a refrigerator.
While terminal parts of the stem are best, a long shoot can be divided into several cuttings. Cuttings are generally 4 to 6 inches long. Use a sharp, thin-bladed pocket knife or sharp pruning shears. If necessary, dip the cutting tool in rubbing alcohol or a mixture of 1 part bleach to 9 parts water to prevent transmitting diseases from infected plant parts to healthy ones.
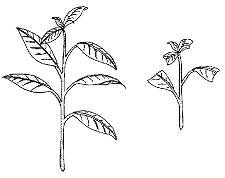
Remove the leaves from the lower one-third to one-half of the cutting (Figure 4). On large-leafed plants, the remaining leaves may be cut in half to reduce water loss and conserve space. Species difficult to root should be wounded.
Treating cuttings with root-promoting compounds can be a valuable tool in stimulating rooting of some plants that might otherwise be difficult to root. Prevent possible contamination of the entire supply of rooting hormone by putting some in a separate container before treating cuttings. Any material that remains after treatment should be discarded and not returned to the original container. Be sure to tap the cuttings to remove excess hormone when using a powder formulation.
The rooting medium should be sterile, low in fertility, and well-drained to provide sufficient aeration. It should also retain enough moisture so that watering does not have to be done too frequently. Materials commonly used are coarse sand, a mixture of one part peat and one part perlite (by volume), or one part peat and one part sand (by volume). Vermiculite by itself is not recommended, because it compacts and tends to hold too much moisture. Media should be watered while being used.
Insert the cuttings one-third to one-half their length into the medium. Maintain the vertical orientation of the stem (do not insert the cuttings upside down). Make sure the buds are pointed up. Space cuttings just far enough apart to allow all leaves to receive sunlight. Water again after inserting the cuttings if the containers or frames are 3 or more inches in depth. Cover the cuttings with plastic and place in indirect light. Avoid direct sun. Keep the medium moist until the cuttings have rooted. Rooting will be improved if the cuttings are misted on a regular basis.
Rooting time varies with the type of cutting, the species being rooted, and environmental conditions. Conifers require more time than broadleaf plants. Late fall or early winter is a good time to root conifers. Once rooted, they may be left in the rooting structure until spring.
Newly rooted cuttings should not be transplanted directly into the landscape. Instead, transplant them into containers or into a bed. Growing them to a larger size before transplanting to a permanent location will increase the chances for survival.
For Further Reading
- Bryant, G. 1995. Propagation Handbook. Stackpole Books: Mechanicsburg, Pennsylvania.
- Dirr, M. A. and C. W. Heuser, Jr. 1987. The Reference Manual of Woody Plant Propagation: From Seed to Tissue Culture. Varsity Press: Athens, Georgia.
- Hartmann, H. T., D. E. Kester, F. T. Davies and R. L. Geneve. 1996. Plant Propagation, Principles and Practices. 6th ed. Prentice Hall: Upper Saddle River, New Jersey.
- McMillan Browse, P. D. A. 1978. Plant Propagation. Simon and Schuster: New York.
- Toogood, A. 1993. Plant Propagation Made Easy. Timber Press: Portland, Oregon.
Publication date: Jan. 31, 1999
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.