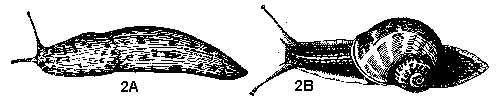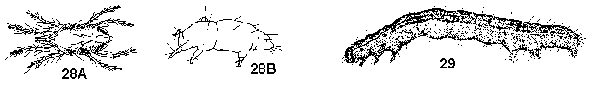Preface
This is the first real revision of a manual first published in 1978, bravely called Insects and Related Pests of Flowers and Foliage Plants. In those innocent days, the Western flower thrips and sweetpotato whitefly were not yet found as floricultural pests in the Eastern United States. The leafminer, Liriomyza trifolii, was then a scourge of greenhouse flowering crops, and aphids were not particularly difficult to control with traditional pesticides. Nine additional entomologists have contributed to the expansion of the first edition, making this a truly regional publication.
A note about the illustrations in this manual. Special effort was made to assure all of the illustrations are all in the public domain. Consequently, no permission is required to use them. Whenever possible, the pests face up or to the left in each illustration. An exception to this rule is found in the Slugs and Snails chapter because the respiratory pore (an important diagnostic character) of slugs is on the right side of the body. Illustrations by: Ponglerd Kooaroon, Suan Van Gieson, Ramona Beshear, Tong-Xian Liu, Mei-Jung Lin, L. L. Deitz, J. J. Davis, H. A. Denmark, J. B. Burch, F. H. Crittenden, Nellie M. Quaintance, O. C. Mohr, James Wilcox, J. R. Baker, California Experiment Station, Connecticut Agricultural Experiment Station and USDA.
Special thanks are extended to Drs. Maurice H. Farrier, NC State; Paul R. Heller, Pennsylvania State University; and Richard L. Miller, Ohio State University, for reviewing the manuscript of the first edition. Their time spent on the manuscript is well appreciated. Thanks also are extended to Katherine Curle who helped with translating much of the early draft into English. The patience of Vicki Grantham, Carmen Sasser, Teresa Snell, Sara Watson, and Stella Nwosu as they prepared the typewritten copies of the manuscript is heartily appreciated. The enthusiastic proofing, guidance on page layout and other editorial duties by Deborah Dunsford is also greatly appreciated.
Introduction
Horticulturists estimate that 1,500 varieties of herbaceous ornamental plants are grown commercially in North Carolina. Ornamentals are grown in commercial greenhouses, commercial fields, professionally maintained landscapes, interior plantscapes, and home grounds, as well as inside homes or in hobby greenhouses.
An amazing variety of insects feed on flowering and foliage plants. Many field, fruit, and vegetable crop pests such as the cabbage looper, corn earworm, diamondback moth, and oblique banded leafroller also feed on ornamentals. Lush growth and sheltered growing conditions make ornamental herbs especially attractive to plant pests.
The routine use of insecticides usually eliminates predaceous insects and mites. However, pests remaining after treatment sometimes tolerate commonly used insecticides. To stay in business, most commercial flower and foliage plant growers must become fairly sophisticated in using various types of pest management practices, insecticide formulations, application equipment, and in rotating insecticides from one chemical group to another.
Commercial growers use a battery of general and restricted-use pesticides. Although amateur gardeners battle the same pests, the most effective chemicals for control are often very expensive or are restricted so that homeowners are not allowed to use them. This puts a moral burden on the commercial grower to try earnestly to eliminate all plant pests from bedding, potted, and cut plants before selling them.
Keys to adults and immatures of the pests referred to above are included in this publication. These keys, plus the color plates and the illustrations in the insect notes, will help you identify the pests of herbaceous ornamental plants. Identification is important to assure proper control measures.
Management of Insect and Related Pests
This manual is designed to augment state Cooperative Extension publications on pest management, not to duplicate them. In no way are the suggestions for safe use and calibrations for proper application in the state Cooperative Extension recommendations to be belittled or ignored. This section on management should be viewed as an expansion of explanations begun in those publications.
Greenhouse
Chemical Control
Total reliance on pesticides for pest management is labor intensive and sometimes very hard on the plants. Although there are numerous practical and philosophical problems with the chemical control of insect and mite pests in the greenhouse, pesticides will remain important in greenhouse pest management at least for the next few years. Control of pests in the greenhouse is often difficult because of lush, sheltered growing conditions. In general, insects, mites, and slugs reproduce more rapidly in warmer temperatures. Also, periodic use of pesticides in greenhouses often reduces parasites and predators of greenhouse pests because the pests tend to be more resistant to pesticides than their predators and parasites.
Application of pesticides is almost essential if one is to stay in the greenhouse profession. Eliminating weeds inside and outside the greenhouse reduces alternate hosts for ornamental plant pests. Screen doors and vents make it harder for moths and beetles to fly in and lay eggs or feed. But, careful as the grower may be, sooner or later an insect or mite will come in on the clothes of workers, on cuttings, in pots, or in soil. Many growers apply pesticides periodically as "insurance" against accidental infestations. Despite these precautions, however, insects or other pests may become established. Some growers treat whenever they discover pests. Growers should survey the plants on a daily or every-other-day basis to guard against extensive damage by insects, mites, or slugs.
Pesticides are applied in greenhouses in almost every conceivable manner. Legal methods are usually the safest! Growers use aerosols, mists, smokes, fogs, dusts, sprays, drenches, and granules – everything but aerial application and backrubs. Aerosols, smokes, mists, and fogs must be applied when the greenhouse is closed (at night or in the winter). Since most pesticides are sensitive to ultraviolet rays, treatments made later in the evening will be more effective. After the waiting period specified on the label has passed, the structure must be well ventilated before workers can safely enter.
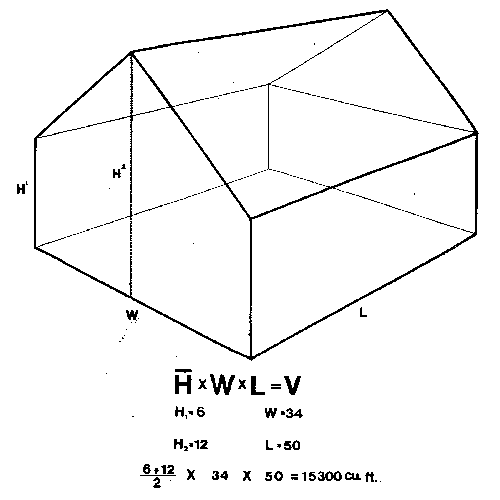
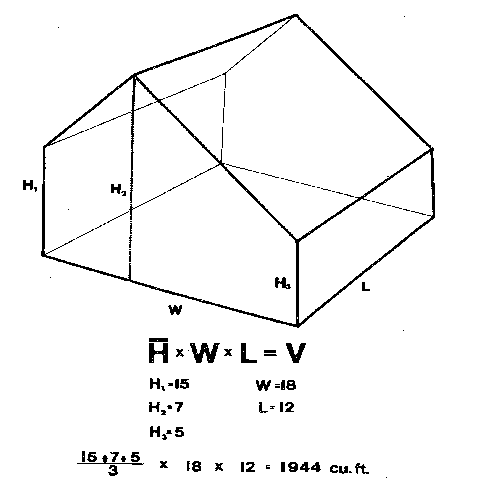
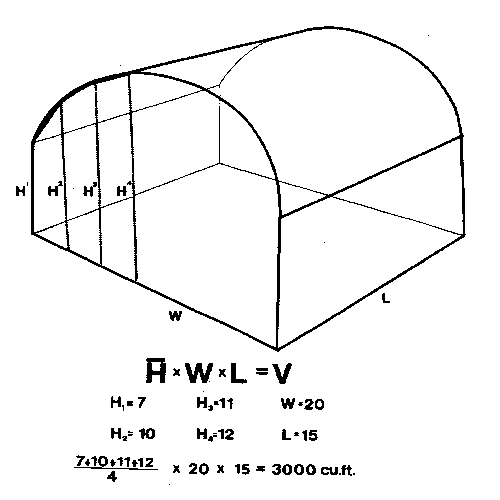
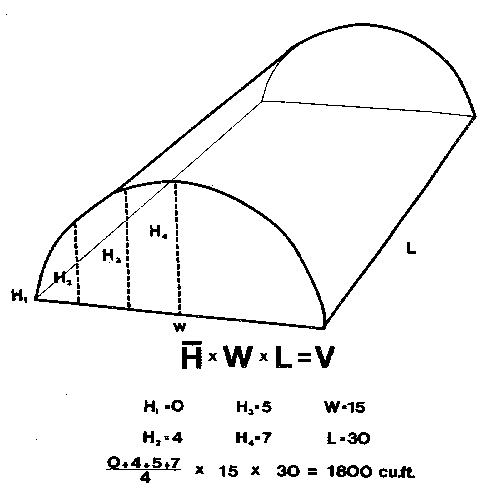
Fogs, smokes, and aerosols are generally applied on a cubic-foot basis. Compute cubic feet by multiplying the area of the floor times the average height of the roof: that is, the length times the width times the average height (Figure 1A). For houses with flat, sloping roofs, add the height at the eave to the height at the highest part, divide by 2, and multiply by the area of the floor (Figure 1B). For houses with rounded roofs, measure the lowest point and the highest point in the middle, then measure the heights at 1⁄4 and 3⁄4 of the distance to the low point. Add these 4 measurements, divide by 4, multiply by the area of the floor, and you have a pretty close estimate of the volume of the house (Figure 1C and Figure 1D). (Although this estimate is a little low, rounded roof houses are usually covered with polyethylene film and are consequently very tight. Thus, applications in these houses do not dissipate as rapidly as applications in fiberglass or glass houses.) Fogs, smokes, and aerosols generally need to be diluted by the air in the greenhouse to avoid damage to the plants.
Place smoke fumigators so that the smoke does not vent directly onto the plant foliage. Growers customarily wear self-contained breathing systems or gas masks when applying fogs, smokes, or aerosols. Some growers who use smoke fumigators determine the number of fumigators needed, based on cubic feet, and light those furthest from the door first. This allows the grower to vacate the house before it becomes dangerously filled with fumes. Two people should always be present when applying toxic substances in the greenhouse. If one person gets accidentally poisoned, the other can drag the victim to safety and call for help. Dusts and sprays are applied with conventional dusting or spraying equipment. Necessary safety clothing must be worn.
Apply granular insecticides with a handheld shaker or some other device that does not grind up the granules. Wear boots, long pants, a long sleeved shirt, rubber gloves, and a respirator. The plants and potting mix should not be handled until the granular pesticide has been washed from the foliage and watered in thoroughly.
Integrated Pest Management
IPM uses all suitable methods to reduce insect and mite populations to the lowest acceptable level. IPM is a complex program as each crop must be considered individually. However, there are a number of basic practices that apply to most greenhouse crops.
Clothing
An effort should be made to avoid carrying insects into the greenhouse by wearing clothes that are brown, red, or black. Do not wear white, yellow, or green, as these colors are attractive to aphids, thrips, whiteflies, leafminers, and darkwinged fungus gnats. Light to dark blues also are attractive to aphids and thrips.
Quarantine
Before any plant material is brought into the greenhouse, it should be thoroughly inspected for insects, mites, and diseases. Furthermore, new plant material should be kept in a separate section for a week or more before such material is incorporated into the production area. Such highly resistant pests as the green peach aphid, western flower thrips, and silverleaf whitefly move readily on plant material. The swapping of insects, mites, and diseases on infested plant material is without doubt the major way resistant thrips, aphids, and whiteflies are transported throughout the greenhouse industry.
Screening
In an experiment with greenhouse screening in California, a crop of chrysanthemums was grown successfully without a single application of pesticides. Exclusion demonstrations at North Carolina State University with the western flower thrips and the silverleaf whitefly have shown that both pests can be significantly excluded by spunbonded and perforated polyethylene screening. With screening, the finer the mesh, the greater is the tendency to restrict air flow into the greenhouse. Polyspun materials cut air flow by a factor of two. The perforated polyethylene screening cut air volume by a factor of five (Figure AA).
Although the pore sizes of some screening materials are large enough that the thrips are capable of wedging through, the screening still excludes many thrips. Evidently, these materials are not recognized by thrips as a suitable substrate to feed on. When the thrips probe the screening they may instinctively resume flight searching for a suitable plant.
Pest Recognition
For proper management, it is important to be able to recognize the various kinds of pests in their various stages of development. Probably the most frequently misidentified pests are shore flies and darkwinged fungus gnats. Shore flies are of little economic consequence in the greenhouse but are very resistant to pesticides. Thus a grower can waste effort and pesticides trying to chemically control shore flies rather than trying to control algae the shore flies are breeding in. Another example of misidentification is the assumption that parasitized green peach aphids are some sort of new 'tan' aphid. Parasitized aphids adhere to the plant fairly tightly, so in spite of repeated applications, these 'tan' aphids seem to be impossible to kill.
Monitoring
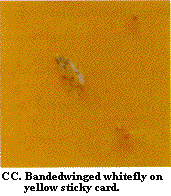
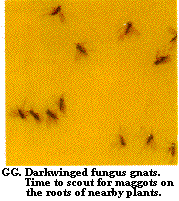
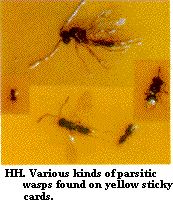
Constant vigilance for insects, mites, and diseases is required for effective pest management. An employee or certain employees should be assigned the responsibility of scouting for insects and other pests on a regular basis (perhaps weekly during the winter and twice weekly during the summer). Written records of where various pests are found should be kept. Pests can be monitored by using yellow and blue sticky cards (Figure BB, Figure CC, Figure DD, Figure EE, Figure FF, Figure GG, Figure HH, Figure II, Figure JJ), by using yellow pan traps, and by examining the foliage, flowers, and occasionally the roots. Light traps outside can be used to monitor for European corn borer, corn earworm, and beet armyworm adults.
Record Keeping
A written log should be kept of pest type, locality, abundance, and all pesticides applied. Such records can be of long-term benefit as many pests tend to appear at about the same time each year. However, the short-term benefits of written records may be greater. Knowing what pests survive a pesticide application alerts the grower to the possibility of poor timing, poor application, or pesticide resistance in the pest population. A change in strategy, application technology, or type of pesticide can be made before the crops are significantly damaged.
Biological Control
Some growers use beneficial organisms for biological control where appropriate. Unfortunately, there are no really effective organisms available for managing the silverleaf whitefly or the western flower thrips. Encarsia formosa parasitic wasps infest silverleaf whiteflies to some degree and Amblyseius predatory mites, used for spider mite suppression, can feed on thrips. Bacterial and nematode organisms can be readily integrated into a traditional pest management scheme, whereas others require a fairly high level of management. Aphytis wasps, Aphidoletes maggots, and green lacewings are available for aphid suppression. Bacillus thuringiensis kurstaki pesticides are available for caterpillars. Bacillus thuringiensis israelensis and Steinernema carpocapsae nematodes suppress darkwinged fungus gnats. Cryptolaemus and Delphastus lady beetles (when available) can be used for mealybugs and whiteflies. Parasitic wasps are available for soft scale management and predaceous mites are available for spider mite suppression. Encarsia formosa parasitic wasps can be used especially for greenhouse whitefly suppression. Except for Bacillus thuringiensis pesticides, the use of biological organisms is usually not compatible with the use of chemical sprays. It is possible to integrate sprays of soaps and oils with Encarsia formosa by timing pesticide applications to coincide with the "black scale stage" of the parasite's development. Also the "brown mummy" stage of aphids infected with Aphytis wasps are resistant to soaps and oils.
Organic Control
Organic growers tend to be wary of relying on pesticides for routine pest management. Most organic growers are highly receptive to the basic integrated pest management practices (screening, biological control, and monitoring). The range of chemicals organic growers can use is limited to those that are certified to be "organic" by various organizations such as the California Certified Organic Farmers. Some of these chemicals work well and others are marginally effective. Finding formulations that are certified as organic and that are actually labeled for greenhouse use is sometimes a problem. Some organic growers in North Carolina use screening to exclude pests and methods such as irrigation to dislodge and destroy mites and aphids or washing the produce by hand at harvest to remove pests.
Organic Chemicals: Soaps, oils, and nicotine sulfate can be used for aphid suppression. Soaps, oils, neem extracts, and pyrethrum sprays and aerosols are moderately toxic to 3 whiteflies. The Bacillus thuringienses kurstaki pesticides and pyrethrum sprays, and aerosols are effective for caterpillar control. Soaps and pyrethrum pesticides suppress mealybugs. Spider mites are susceptible to soaps and oils. Pyrethrum sprays and aerosols help suppress thrips.
Commercial Indoor Landscape
Commercial indoor landscape accounts are perhaps the most difficult areas in which to attempt pest control. Interior plantscapes extend from public conservatories to extensive plantings in homes, hotels, office buildings, restaurants, shopping malls, hospitals, schools, and other environmentally sensitive areas. The use of insecticides in many of these areas is often greatly restricted because of the sensitivity of the surroundings. In addition, few chemicals are cleared for ornamental plant use in public areas, and public prejudice against pesticide odors can prevent application of pesticides in many situations.
The first line of defense against ornamental plant pests is sanitation and quarantine. Taking steps to prevent pest problems is well worth the effort. Inspect plants thoroughly and only place insect-free plants into an indoor landscape. If possible, when insect infestations are first found, isolate infested plants to prevent spread of the infestation.
Interest in biological control in indoor landscapes has expanded in recent years because of restrictions placed on interior plantscape pesticide application, pesticide costs, poor control with pesticide products, phytotoxicity, and potential human health hazards. To be successful at managing pest problems with biological control agents requires a knowledge of the biology of the pest species, the biological control agent(s), and a great deal of time and commitment. Biological control systems do not look after themselves.
Thorough, timely applications of properly labeled pesticides are another important aspect of an integrated pest control program. Rotating infested plants back into a greenhouse where they can be treated thoroughly and revived in vigor before being returned to the indoor landscape is a sound practice but is often not practical. Treat commercial landscape areas at night, on weekends, or when a minimum number of people are present. The public should not be allowed in the vicinity of treated plants until the pesticide residue on the foliage has dried completely.
Commercial Field Grown Flowers
This is the sector of the ornamental plant industry that most closely parallels agricultural crop production in methods and equipment. Tractor-drawn spray rigs or mist blowers are often used for pest control. A broad range of chemicals is cleared for use on field-grown flowers. However, the number of insect and mite pests that can be tolerated is very low. Vigilance must be maintained to prevent extensive damage by ornamental plant insect, mite, and slug pests.
Key to Orders and Groups of Pests
- Body and antennae unsegmented, contractile, slimy; entire lower surface modified as a sort of gliding foot; breathe through single, closable opening on right side of body (Figure 2A, 2B) - SLUGS AND SNAILS
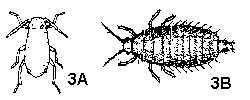
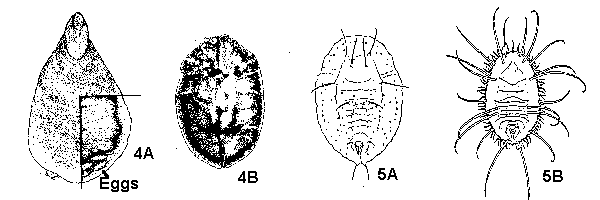
Body and appendages segmented (Figure 3A, 3B), or if apparently not segmented, then specimens either permanently attached to plants (scales, Figure 4A, 4B and whiteflies, Figure 5A, 5B) or less than 1 mm long (mites, Figure 6A, 6B) - GO TO 2 - Some combination of the following: wings; three or four pairs of segmented legs; a pair of large compound eyes; a pair of well-developed antennae; body larger, darker and/or harder than that of the immature. (Adult and immatures of mites, aphids, mealybugs, and scales are very similar except for size and reproductive maturity and can be difficult to distinguish) - ADULTS
No wings; some of the following present or absent: segmented thoracic legs; fleshy abdominal prolegs; eyes; antennae; body larviform (caterpillar, grub, or maggot) or at least smaller, paler, and softer than adult - IMMATURES
Key to Adults
- Specimen permanently attached to leaves or stems, may be hidden under shell or wax secreted by specimen (Figure 4A, 4B and Figure 5A, 5B) - SCALES AND IMMATURE WHITEFLIES
Specimen mobile, not permanently attached to vegetation - GO TO 2 - Body unsegmented, usually less than 1 mm long; 4 pairs of segmented thoracic legs; wings, compound eyes, and antennae always absent (Figure 6A, 6B) - MITES
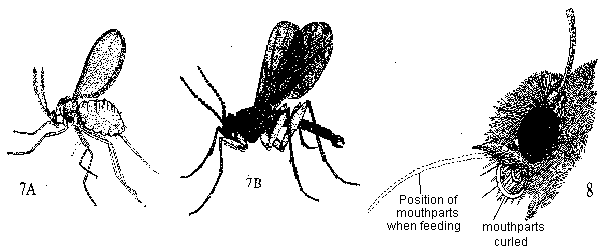
- With 1 pair of transparent front wings; hind wings reduced to knob-like halters (Figure 7A, 7B) - FLIES
With 2 pairs of wings or none, front wings may be shell-like, hiding the hind wings - GO TO 4 - Body densely covered with scales and hairs; mouthparts in form of slender, unsegmented tube that is coiled and concealed under head (Figure 8) - MOTHS AND BUTTERFLIES
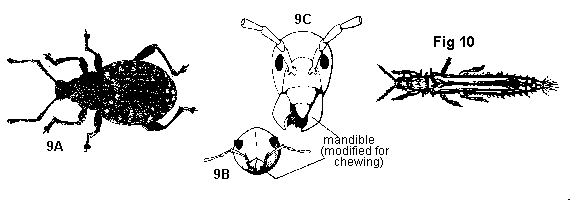
Scales and hairs, if present, sparse and not concealing body surface; mouthparts different - GO TO 5 - Front wings in form of a hard or leathery shell meeting at midline but not overlapping, covering the transparent folded hind wings; mouthparts of chewing type, mandibles on head or at tip of unsegmented snout (Figure 9A, 9B, 9C) - BEETLES
Front wings, if present, either transparent, flexible, or overlapping when closed; mouthparts a segmented beak or cone for piercing and sucking - GO TO 6 - Tiny, slender insects more than twice as long as wide, usually 1 to 2 mm long; wings folded flat on back when closed, overlapping, much narrower than width of body, each bearing a long fringe of marginal hairs (Figure 10) - THRIPS
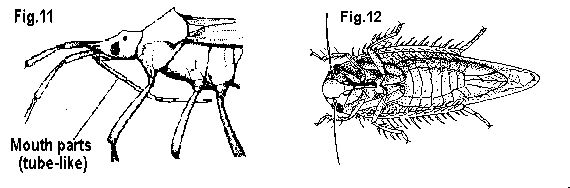
Not fitting this description - GO TO 7 - Mouthparts a segmented, hinged beak attached to underside of head anterior to eyes (Figure 11); antenna of four or five distinct segments; front wings folded flat on back, usually (except lace bugs) leathery at base and membranous toward tips, tips overlapping - BUGS
Mouthparts a short beak attached to head under eyes or even between front legs; antennae and wings variable (Figure 12) - GO TO 8 - Antennae distinct, segmented, arising from head in front of eyes; insects crawl or fly but don't jump - GO TO 9
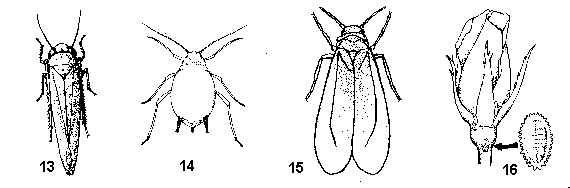
Antennae slender, hairlike, arising from knobs beneath eyes; hind legs long, spiny, adapted for jumping; alert, active insects (Figure 13) - LEAFHOPPERS - A pair of tubelike cornicles on abdomen (Figure 14) - APHIDS
Cornicles absent - GO TO 10 - Fully winged, tiny, whitish, active insects with large eyes; found on undersides of leaves among their immobile immatures; fly in spiral pattern when disturbed (Figure 15) - WHITEFLIES
Wingless, slow-moving, oval insects; body pinkish or grayish under a coat of powdery white wax, often with filaments and tufts of hairlike wax; usually concealed on various parts of plants (Figure 16) - MEALYBUGS
Key to Immatures
- Becoming permanently attached to leaves or stems, may be hidden under shell or wax secreted by specimen (Figure 17A, 17B and Figure 18A, 18B) - SCALES AND WHITEFLIES
Mobile, or capable of crawling when prodded - GO TO 2 - Lack of legs - GO TO 3
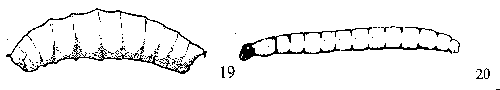
Possesses legs of some sort - GO TO 5 - Headless, with a pair of retractable mouth hooks, a maggot (Figure 19) - FLY MAGGOTS
Has a distinct head capsule (Figure 20, Figure 21) - GO TO 4 - Slender maggot with shiny black head capsule, found in decaying plant tissues and in fungi (Figure 20) - DARKWINGED FUNGUS GNATS
Stout-bodied grub with orange or brown head capsule, in soil around roots or boring in healthy plant tissue (Figure 21) - WEEVILS AND OTHER BEETLES - Tiny, body unsegmented, less than 0.5 mm long; 3 or 4 pairs of segmented legs present; eyes and antennae always absent (Figure 22) - MITES
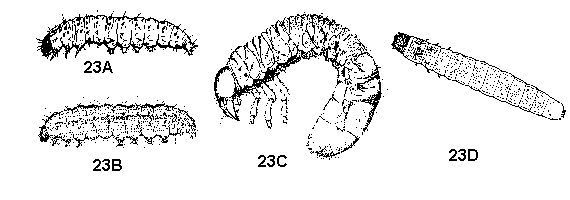
Body segmented, usually longer than 0.5 mm; only 3 pairs of segmented legs; eyes and antennae usually present but may be small or obscure - GO TO 6 - Immature a caterpillar or grub, with chewing mouthparts; looking very different from, and not found with, the adult (Figure 23A, 23B, 23C, 23D) - GO TO 7
Immature a nymph with a beak or cone-shaped mouthparts: similar to, and often found in groups with adults - GO TO 8 - Immatures with 3 or 4 pairs of fleshy abdominal prolegs and 1 pair of anal prolegs; some species can spin silk, which they use to tie foliage together, line burrows and spin cocoons (Figure 23A, 23B) - CATERPILLARS
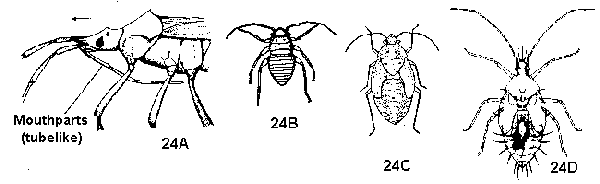
Immatures lacking prolegs; cannot spin silk (Figure 23C, 23D) - WHITE GRUBS, WIREWORMS, AND OTHER BEETLES - Mouthparts a segmented, hinged beak attached to underside of head anterior to eyes; antennae of 4 distinct segments; compound eyes well developed; stout-bodied, slender-legged, usually active and solitary nymphs (except lace bugs, which are spiny) (Figure 24A, 24B, 24C, 24D) - BUGS
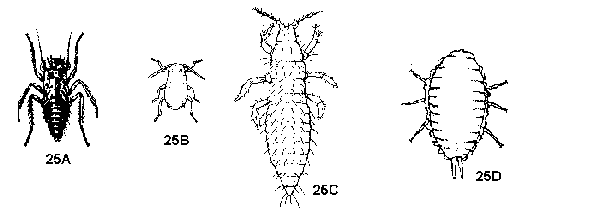
Mouthparts a short beak attached to underside of head beneath eyes or between front legs; antennae and eyes variable; body form variable (Figure 25) - GO TO 9 - Active, alert insects, solitary on foliage and stems; hind legs long, spiny, can jump; antennae hairlike, arising from head underneath eyes and above beak (Figure 25A) - LEAFHOPPERS
Less active, often found in groups or concealed; hind legs not adapted for jumping; antennae arising from head in front of eyes - GO TO 10 - Possessing a pair of tubelike cornicles on abdomen; usually found in clusters or colonies on leaves, new shoots, nymphs and adults together (Figure 25B) - APHIDS
Lacking cornicles - GO TO 11 - Slender insects, more than twice as long as wide; usually pale yellow or red, lacking a coating of powdery or filamentous wax; eyes visible; mouthparts concealed in a cone on part of head (Figure 25C) - THRIPS
Oval or rounded insects, less than twice as wide as long; body pinkish or grayish beneath a coating of powdery or filamentous wax; eyes lacking; mouthparts a short beak between front legs (Figure 25D) - MEALYBUGS
Key to Blossom and Leaf Feeders
- Body and antennae unsegmented, contractile, slimy, legless; entire ventral surface modified as a sort of gliding foot; breathe through single, closable opening on right side of body (Figure 26A, 26B) - SLUGS AND SNAILS
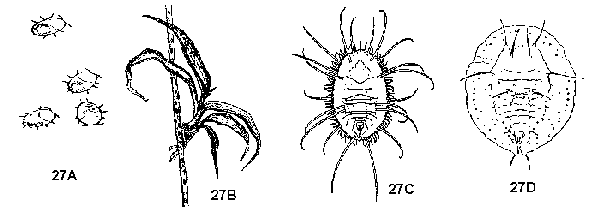
Body and appendages segmented, or if apparently not segmented. then specimens either attached to plants or less than 1 mm long - GO TO 2 - Specimens attached to plant, rarely if ever moving (Figure 27A, 27B, 27C, 27D) - SCALES AND IMMATURE WHITEFLIES
Specimens readily mobile - GO TO 3 - Tiny, 1 mm or less long; 3 or 4 pairs of segmented thoracic legs; antennae, compound eyes, and wings always absent; body unsegmented (Figure 28A, 28B) - MITES
Usually larger; 3 pairs of segmented thoracic legs; antennae, compound eyes, and wings (adults) usually present although they may be reduced or hidden; body segmented - GO TO 4 - Body elongate, soft; head capsule smooth and rounded, hard; eyes and antennae reduced; 3 pairs of thoracic legs plus 1 or more pairs of fleshy abdominal prolegs and 1 pair of fleshy anal prolegs (Figure 29) - CATERPILLARS
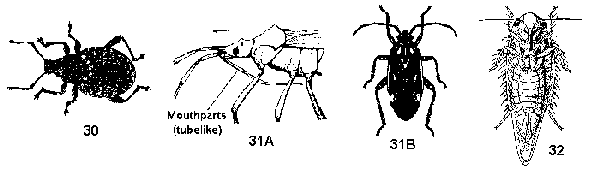
Not a caterpillar, abdominal and anal prolegs absent - GO TO 5 - Mouthparts are chewing mandibles; body hard; front wings not overlapping, meeting at midline of body, modified into a shell that covers the transparent, folded hind wings; antennae may be elbowed, clubbed, or otherwise modified (Figure 30) - BEETLES
Mouthparts beaklike or conelike, adapted for piercing and sucking; body leathery or soft; front wings not like a shell, may partly or completely overlap at rest; antennae (except leafhoppers) a simple, straight series of segments - GO TO 6 - Mouthparts an elongate three or four-segmented beak that arises in front of eyes on underside of head (Figure 31A); large compound eyes always present; antennae of 4 or 5 distinct segments; front wings of 2 textures, leathery near base and membranous at tips, tips overlapping (Figure 31B) - BUGS
Mouthparts a beak or cone, arising beneath eyes or (apparently) between first pair of legs (Figure 32); eyes and antennae variable; front wing of uniform texture - GO TO 7 - Slender insects, more than twice as long as wide - GO TO 8
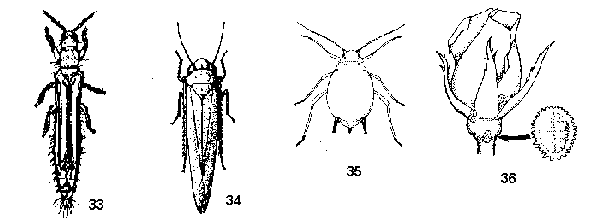
Oval or rounded insects, less than twice as long as wide - GO TO 9 - Tiny yellow, brown or black insects, 1 to 2 mm long; antennae long, projecting from front of head; hind legs short, not modified for jumping; front wings (if present lying flat on back when closed, completely overlapping, each wing with a fringe of long hairs on its margins (Figure 33) - THRIPS
Usually larger than 1 to 2 mm when mature; variously colored and patterned, often green; hind legs long, modified for jumping; antennae short, hairlike, arising under eyes; front wings not lying flat on back or overlapping when closed, lacking fringe (Figure 34) - LEAFHOPPERS - With a pair of tubelike cornicles on abdomen; large compound eyes present; legs long and slender; body variously colored and patterned (Figure 35) - APHIDS
Cornicles and compound eyes absent; legs short, not slender; body pinkish or grayish; often covered with a white, waxy bloom (Figure 36) - MEALYBUGS
Key to Stem Borers
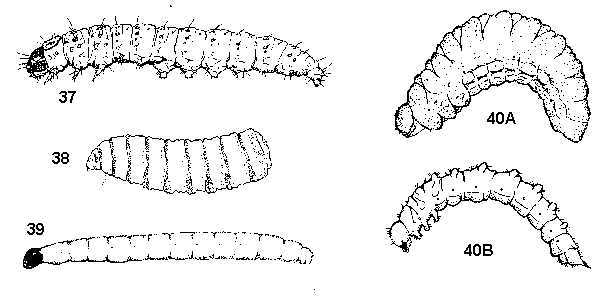
- With 3 pairs of short, segmented thoracic legs, plus 1 or more pairs of fleshy abdominal prolegs and 1 pair of fleshy anal prolegs (Figure 37) - CATERPILLARS
Abdominal and anal prolegs absent - GO TO 2 - A distinct, hard, pigmented head capsule present, mouthparts are chewing mandibles - GO TO 3
No distinct head capsule, mouthparts are pair of retractile, downward curving hooks (Figure 38) - MAGGOTS - Head capsule shiny black; body slender, smooth, soft, white; thoracic legs absent (Figure 39) - DARKWINGED FUNGUS GNATS
Head capsule orange to brown; body variable; thoracic legs may be present (Figure 40A, 40B) - BEETLES
Key to Root Feeders
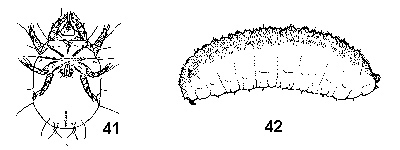
- Tiny, slow moving, 1 mm or less long; 3 or 4 pairs of segmented thoracic legs; head and mouthparts cone shaped, eyes and antennae absent; body bulbous, unsegmented, with scattered long hairs; found in decaying or moldy vegetation (Figure 41) - MITES
Larger; 3 pairs of segmented thoracic legs or all legs absent; body segmented; caterpillars, grubs, and maggots - GO TO 2 - A distinct head present; mouthparts are chewing mandibles or a sucking beak - GO TO 3
No head capsule; mouthparts are a pair of retractile, downward curving hooks (Figure 42) - MAGGOTS - All legs present - GO TO 4
Some legs present - GO TO 5 - Head capsule shiny black; body slender (Figure 39) - DARKWINGED FUNGUS GNATS
Head capsule orange to brown; body usually stout (Figure 40A and Figure 43) - WEEVILS, LEAF BEETLES - With three pairs of segmented thoracic legs, plus 4 pairs of fleshy abdominal prolegs and 1 pair of fleshy anal prolegs (Figure 37) - CATERPILLARS
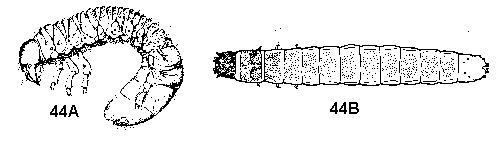
Prolegs absent - GO TO 6 - Head capsule distinct from body; mouthparts are chewing mandibles; body more than twice as long as wide; white, yellow, or orange to brown in color; no wax (Figure 44A, 44B) - WHITE GRUBS AND WIREWORMS
Head not distinct from body; mouthparts slender, short beak; body oval or rounded, usually less than twice as long as wide; pale pink or gray under a coating of powdery wax (Figure 45) - MEALYBUGS
Home Grounds and Home Dwellings
Homeowners have difficulty in pest management of their grounds, in part due to the great diversity of ornamental plants. Homeowners are often not aware that many of these plants have insect and mite pests. Injury caused by insects and related pests is another source of potential confusion if the insect or mite is not available to help in the diagnosis. When they are willing to educate themselves to recognize problems and to frequently inspect their ornamentals, homeowners can limit the number and scope of their pest problems.
Home gardeners have an adequate selection of pesticides labeled for home use, especially for home ornamentals. A variety of application equipment options are available to the amateur gardener. Probably the two most popular applicators are dusters and hose-end sprayers. Dusts require no mixing and are often applied directly from the purchased container, which has a perforated top. Plunger dusters and bulb dusters are still in use, but not as popular. These types have a reservoir that is filled with the dust. Dusts offer quick and convenient application, but are best applied when there is little wind to reduce drift. Hose-end sprayers use water pressure to siphon, dilute, and deliver the pesticide to its destination. Pesticide concentrate is placed in the reservoir of the hose-end sprayer and water added based on the volume of spray needed. Length of hose limits use of hose-end sprayers on large properties. The hose-end sprayer reservoir must be rinsed after each use.
Compressed air sprayers are available in several sizes and prices and are popular in the home garden. These sprayers use air pressure to deliver the diluted pesticide through the spray nozzle to the target. The nozzle is often adjustable, from a coarse to fine spray pattern. Spray coverage is more uniform with compressed air sprayers than with dusters and hose-end sprayers. Pesticides must be diluted according to label instructions, and sprayers must be cleaned after each use.
Ready-to-use pesticides and sprayers are the newest application technology. Pesticides are sold premixed, and the reservoir is connected to a plastic nozzle. The system is a closed one, with no mixing or cleaning necessary. Pressure is applied by a trigger. The system is adequate for small areas, but would not provide coverage to larger plantings.
Sprayers mounted on garden-type tractors similar to those available for farm use can be used on larger properties. Some models use the tractor battery to power the sprayer and others use a power-take-off system. As with other sprayers, proper mixing of pesticides and thorough cleaning of the equipment after use are required.
Publication date: Jan. 1, 1994
AG-136
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.