Key to Boxwood Pests
Approximately 3,600,000 boxwoods are produced each year by nurserymen in the southeastern United States. Although handsome and stately plants, boxwoods are sometimes difficult to grow. In the wrong situation, boxwoods are susceptible to leafminers, wax scales, and other insects as well as spider mites, nematodes, and various root diseases.
- Boxwood leafminer – The leaf's lower surface appears "puffy" or blistered. Heavily infested plants have distorted and discolored leaves.
- Boxwood psyllid – New growth is distorted and cup shaped. Pale-green insects (up to 3 mm long) feed inside distorted growth and secrete a fluffy, waxy covering; or small, green, leafhopper-like insects jump or fly onto foliage.
- Japanese wax scale – White, waxy blobs up to 6 mm in diameter appear on the limbs and twigs. They are often accompanied by a sooty mold.
- Spider mites – The leaves are speckled with tiny, pale dots. The whole shrub may appear off-color, gray, or bronzed.
- Twospotted spider mite – Damage appears as aggregates of single dots. The eggs are round and often reddish.
- Boxwood spider mite – Damage appears as tiny lines or "hen scratches" on the leaf surface. The eggs are flattened and yellowish.
Boxwood Leafminer
Boxwood leafminer, Monarthropalpus buxi (Laboulbene), Cecidomyiidae, DIPTERA
DESCRIPTION
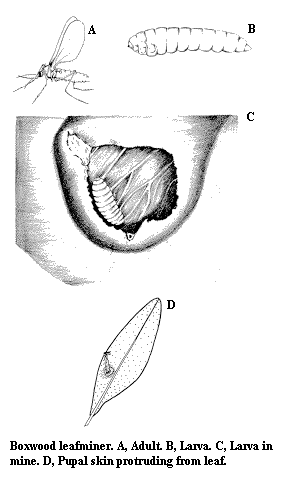
Adult – The mature boxwood leafminer is a yellow to orange-red fly. Mosquito like but small (2.5 mm), it can often be observed swarming around boxwoods during the time weigelas are in bloom (Figure K).
Larva – The larva is a small, whitish to lemon-yellow maggot up to 3 mm in length (Figure L).
Pupa – The elongate, 3-mm-long pupa is whitish to dark yellow. Close examination reveals legs and wings appressed to the body surface.
BIOLOGY
Distribution – The boxwood leafminer is probably the most commonly reported pest of boxwoods. Imported from Europe, this small fly is a greater pest in the Mountains and Piedmont than in the Coastal Plain. However, infestations can occur wherever boxwoods are grown.
Host Plants – All boxwoods may be infested, but more slowly growing English varieties are less susceptible than American varieties.
Damage – Mining in the foliage, this pest causes the formation of small blisters on the undersurfaces of leaves (Figure M). Infested leaves usually become yellowish and are smaller than uninfested leaves. As a result, heavily infested plants assume an unthrifty appearance.
Life History – Adult flies insert their eggs into the leaves' upper surfaces. Tiny larvae hatch and mine into the leaves as they feed. The leaves first acquire a water-soaked appearance at the feeding site. Soon, blisters develop on the lower leaf surface; one to several larvae may develop on a single leaf. The leafminers spend the winter in the blisters as larvae. In spring, the blisters develop a translucent "window" through which pupae protrude from the lower leaf surface. Adult flies emerge from the pupae over a 2-week period in early spring shortly after the boxwoods have put out their new growth. The adult flies live only a few days. Only one generation occurs each year.
CONTROL
For specific chemical controls, see the current state extension recommendations.
Boxwood Psyllid
Boxwood psyllid, Psylla buxi (Linnaeus), Psyllidae, HEMIPTERA
DESCRIPTION
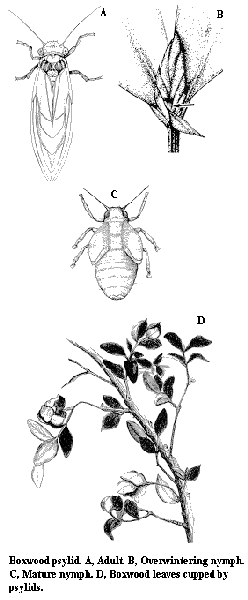
Adult – This small (3-mm-long), grayish-green sucking insect has transparent wings and resembles a miniature cicada (Figure N).
Egg –The tiny, orange egg is spindle shaped.
Nymph – The flat, green- and brown-mottled nymph is covered with whitish, waxy filaments. The nymph is wingless and smaller than the adult (Figure O).
BIOLOGY
Distribution – Boxwood psyllids seem to occur wherever boxwoods are grown. In the United States, however, they are most common in temperate areas.
Host Plants – Boxwood is the only known host of this pest. Although both American and English varieties are attacked, American boxwoods are more likely to be severely infested.
Damage – Psyllid nymphs extract sap from buds and young foliage. As a result, terminal leaves of infested plants become cupped and twig growth may be checked (Figure P). Since the boxwood psyllid completes its single annual generation early in the growing season, plants tend to outgrow their injury by midsummer.
Life History – Boxwood psyllids overwinter as first-instar nymphs still within their orange egg shells. In spring as buds begin to grow and leaves unfold, the nymphs hatch from the eggs. They immediately begin to suck sap from new leaves. As their feeding causes leaves to curl, the nymphs become concealed and protected. After developing through several instars, psyllid nymphs molt into adults in May or early June. Although adults continue to feed, they are not as damaging as nymphs. In July or August, female adults deposit one to seven eggs under each bud scale. First-instar nymphs develop within the egg before winter but do not emerge until spring. Only one generation occurs each year.
CONTROL
Insecticides are available for control of boxwood psyllids. Infested plants should be treated when new growth appears, and applications should be repeated as necessary. For specific chemical controls, see the current state extension recommendations.
Boxwood Spider Mite
Boxwood spider mite, Eurytetranychus buxi (German), Tetranychidae, PROSTIGMATA
DESCRIPTION
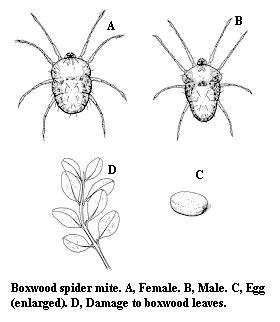
Adult – The adult is green to yellowish brown. The body is ovate in females and tapering in males. It is slightly less than .375 mm long and has eight legs.
Egg – The lemon-yellow egg is flattened on the bottom and slightly flattened on top.
Larva – The larva is green. Except for its small size and six legs, it resembles the adult.
Nymph – The green nymph is similar to the adult except for size.
BIOLOGY
Distribution – This mite has been reported in North Carolina, Virginia, Georgia, Connecticut, Michigan, Oregon, and California.
Host Plants – Boxwoods are the only known host for the boxwood spider mite.
Damage – All stages of mites feed on the upper and lower surfaces of the leaves. As they feed, they apparently inject a toxic saliva, which causes small, yellow, scratchlike spots to form on the upper leaf surfaces. New plant growth seems particularly susceptible to attack. Twospotted spider mites and other spider mites, on the other hand, usually cause tiny chlorotic stipples of discrete dots.
Life History – Overwintering eggs are laid in September and October. In midspring the eggs hatch into six-legged larvae, which crawl about and feed freely for about 3 days. After a resting stage (about 1 day), the larvae molt into eight-legged first nymphs. They feed for about 4 days and then go into a resting stage (about 2 days). After that period, the second nymphs emerge to feed for about 4 days. The final resting stage lasts about 4 days, and then the adults emerge. Mating takes place immediately. Within hours a female may start laying eggs, usually 25 to 30. Because the entire life cycle takes from 18 to 21 days, there are at least eight generations per year.
CONTROL
For specific chemical controls, see the current state extension recommendations.
Japanese Wax Scale
Japanese wax scale, Ceroplastes ceriferus (Fabricius), Coccidae, HEMIPTERA
DESCRIPTION
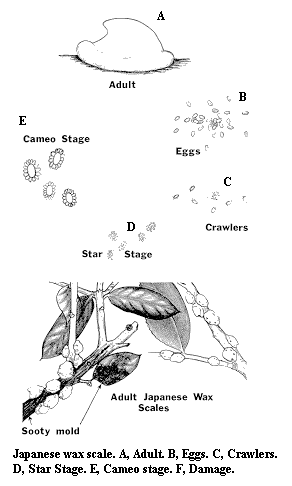
Adult – The brownish-purple female has tiny legs and a thick covering of sticky wax. There are no males.
Egg – Ovoid and pale purple, the egg resembles purple pollen when shaken onto a white surface.
Crawler – Each crawler is flattened and tiny. In a mass, newly hatched crawlers appear rusty red.
Nymph – After molting, each scale secretes wax in tufts, forming a star.
BIOLOGY
Distribution – The Japanese wax scale is found from Florida to Virginia and Maryland.
Host Plants – Japanese wax scales have been reported on azalea, blueberry, camellia, Chinese elm, citrus, fig, eugenia, gumbo-limbo, Chinese holly, yaupon, jasmine, mulberry, pear, persimmon, plum, quince, sapodilla, Turks cap, and other plants.
Damage – A severe infestation of Japanese wax scale detracts from the host plant's appearance because of the many white scales and the copious honeydew that they excrete. A black fungus called sooty mold grows in the honeydew, further disfiguring the host plant.
Life History – Japanese wax scales begin to lay eggs in March, each scale laying from 1,000 to 2,000 eggs. By late May, tiny crawlers hatch and move about, searching for a place to feed. Feeding occurs along the twigs and leaf midribs of numerous woody plants. Once the crawlers insert their sucking mouthparts into the host plant, they are immovable. They then secrete the waxy covering from which they derive their name. The young scales mature throughout the summer, producing more waxy covering and becoming increasingly tolerant to pesticides. They overwinter as adults.
CONTROL
Handpicking scales in winter (if practical) is an effective control measure. Since they may lay their eggs apart from their host plants, the scales should be destroyed after removal. The Japanese wax scale seems to be one of the most difficult ornamental plant pests to control. However because there is only one generation per year, applying pesticides to the crawler stage in early June will give virtually complete control. Applications of any pesticide must be repeated because the crawlers emerge from the female's protective wax coating for 2 or 3 weeks and because recommended pesticides grow ineffective in the environment. For specific controls, see the current state extension recommendations.
Twospotted Spider Mite
Twospotted spider mite, Tetranychus urticae Koch, Tetranychidae, PROSTIGMATA
DESCRIPTION
Adult – The eight-legged adult can be rusty green, greenish amber, or yellow. The overwintering female, however, is orange. Usually having two (sometimes four) black spots on top, the adult is about .375 mm long (Figure HH).
Egg – The spherical egg ranges in color from transparent and colorless to opaque straw yellow.
Larva – The six-legged larva is colorless to pale green or yellow.
Nymph – Similar to the adult except in size, the pale green to brownish-green nymph has eight legs. Large spots of black may develop on each side.
BIOLOGY
Distribution – Twospotted spider mites are widely distributed in the United States.
Host Plants – Twospotted spider mites have been reported on over 180 host plants, which include over 100 cultivated species. Violets, chickweed, pokeweed, wild mustard, and blackberry are common foci from which infestations develop on nearby crops.
Damage – Twospotted spider mites pierce the epidermis of the host plant leaf with their sharp, slender mouthparts. When they extract the sap, the mesophyll tissue of the leaf collapses in the area of the puncture. Soon a chlorotic spot forms at each feeding site. After a heavy attack, an entire plant may become yellowed, bronzed, or killed completely (Figure II). The mites may spin so much webbing over the plant that it becomes entirely covered.
Life History – Twospotted spider mites occur as important pests on more crops than any insect pest in the Southeast. Though insects and mites are in a group called the Arthropoda (meaning jointed foot), because jointed legs are common to both, spider mites are not actually insects. Being more closely related to spiders, they derive their name from the thin web that some species spin. In the Southeast, twospotted spider mites overwinter as adults in the soil or on weed hosts such as violets and hollyhocks. In mild winter weather, twospotted spider mites continue to feed and lay eggs, although development in the winter is much slower than in the summer. Six legged larvae hatch from the eggs. They develop into eight legged nymphs, which pass through two nymphal stages. After each larval and nymphal stage, there is a resting stage. The adults mate soon after emerging from the last resting stage, and in warm weather the females soon lay eggs. Each female may lay over 100 eggs in her life and up to 19 eggs per day. Development is most rapid during hot, dry weather. A single generation may require as many as 20 or as few as 5 days to reach adulthood and begin producing offspring.
CONTROL
The resting stages and eggs of the twospotted spider mite are more tolerant to pesticides than the motile forms. Consequently, a second application of pesticide may be necessary at a 4- or 5-day interval in hot weather (a 7- to 10 day interval in cool weather) to kill those mites that may have survived the first application. For specific chemical controls, see the current state extension recommendations.
Publication date: Jan. 1, 1993
Reviewed/Revised: Aug. 9, 2024
AG-189
Other Publications in Insect and Related Pests of Shrubs
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.