Introduction
Nitrogen (N) is the most important nutrient for ensuring a healthy tobacco crop that produces high quality leaf. Accordingly, the majority of this research focuses on organic N fertilizer sources. North Carolina soils tend to have sufficient levels of legacy phosphorus (P) and potassium (K) for flue-cured tobacco; however, the producers must frequently test soils for essential nutrient concentrations and follow the technical recommendations to keep all the essential nutrients at adequate levels for tobacco.
Greenhouse Management
A tobacco float system is a semi-hydroponic system in which seedling nutrients are applied to the float system solution. Seedlings are grown on a soilless media complex that contains trace amounts of agricultural lime and fertilizer. The limited quantity of fertilizer within the media blend is not enough to promote sufficient seedling growth and development; therefore, supplemental nutrients must be added to the underlying water solution seven to ten days after floating and again roughly three weeks later. It takes 50 to 60 days for seedlings to reach transplant size. Just as soil must be tested to determine if any nutrient or fertility issues must be addressed, float system water should be tested for pH and alkalinity/bicarbonate levels.
Peruvian seabird guano (SG) is a natural product mined from SG deposits commonly found off the Pacific coast of South America and a commonly used fertilizer in organic tobacco float bed systems. Various formulations and brands of SG exist; however, those most commonly used by tobacco producers are high in organic N and P and low in K (for example, Sunleaves® 12-11-2). Many producers have expressed concern with the use of fertilizer sources high in organic N due to negative effects on seedling development, specifically as urea (uric acid) is released from the nutrient source. In addition, as producers add SG to the float water at rates designed to supply sufficient N (typically 125–150 ppm N), they often oversupply P (3x) and undersupply K (–6x). In addition, tobacco float beds fertilized with SG often contain high concentrations of bicarbonate (HCO3-), which can increase water pH, limit availability of some plant nutrients, and reduce seedling growth and vigor.
To improve nutrient recommendations for organic tobacco producers, research was designed to evaluate three organic N programs that might (1) provide sufficient N for seedling growth, (2) limit phosphorus exposure, and (3) reduce bicarbonate concentrations (prevent high float water pH).
Three organic N sources evaluated were 100% SG; 100% sodium nitrate (SN) (SQM Allganic® 16-0-0); and 50% SG + 50% SN. Sodium nitrate fertilizer is mined in the Andes mountains in South America, contains 100% nitrate-N, and is listed as an Organic Materials Review Institute (OMRI) fertilizer. Treatments were supplemented with OMRI-listed water-soluble potassium sulfate (SQM Allganic® 0-0-52). Each fertility program was designed to provide 125 ppm of N and K in all programs, while P varied (0–115 ppm). Each treatment was compared to a nonorganic, homogenized water-soluble fertilizer source (SQM 16-5-16). Fertilizer sources were added to each float bed at 9 and 21 days after seeding (floating). Prior to treatment application, each source was added to a 16 fl. oz. bottle and dissolved in hot water (122°F). Seabird guano was ground to pass through a 1-mm sieve to increase solubility. Total seedlings, usable seedlings, seedling stem height, and stem diameter were measured and recorded at the conclusion of the study. Table 1 shows the complete list of treatments and the nutrients supplied by each fertilizer program.
|
Fertilizer Programa |
N |
P |
K |
Ca |
Quantity (oz/100 gal water) |
|
(ppm) |
|||||
|
16-5-16 |
125 |
40 |
125 |
0 |
10.4 |
|
SG + PS |
125 |
115 |
20 (SG) + 105 (PS) |
0 |
13.9 + 2.7 |
|
SN + PS |
125 |
0 |
125 (PS) |
0 |
10.4 + 3.2 |
|
SG + SN + PS |
44(SG) + 81(SN) |
40 |
7 (SG) + 118 (PS) |
0 |
4.9 + 6.8 + 3.0 |
| a SG: seabird guano; PS: K sulfate; SN: sodium nitrate. | |||||
Treatments containing SN as the sole source of N failed to produce usable seedlings due to the absence of P in the selected fertilizer program (Table 2) and the low P (<1.0 ppm) content of the soilless media source. Seedling growth and development were acceptable in treatments composed of SG or SG+SN and were similar to that of 16-5-16 (Table 2).
|
Fertilizer Programb |
Total Plants (%) |
Usable Plants (%) |
Stem Diameter (mm) |
Stem Height (cm) |
|
16-5-16 |
88 a |
79 bc |
3.30 a |
6.62 a |
|
SG + PS |
90 a |
78 c |
2.76 b |
5.62 b |
|
SN + PSc |
|
|
|
|
|
SG + SN + PS |
91 a |
85 ab |
3.48 a |
6.27 ab |
| a Treatment means followed by the same letter within the same column are not significantly different. b SG: Seabird Guano; PS: potassium sulfate; SN: sodium nitrate. c Treatment did not produce usable transplants; therefore, data are excluded from the analysis. |
||||
Should calcium deficiency develop, producers are encouraged to use OMRI-listed sources of gypsum. The use of lime is discouraged, as it may increase the solution pH to a level that limits availability of other nutrients—in much the same way bicarbonates would.
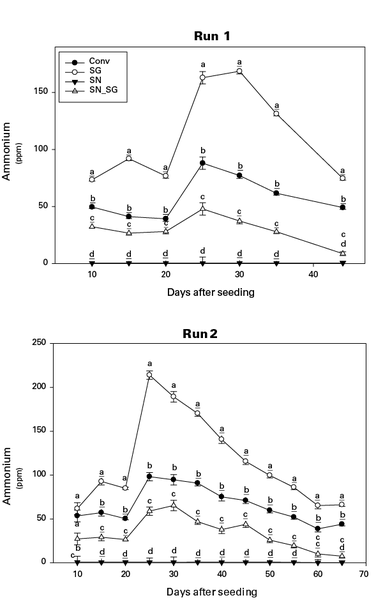
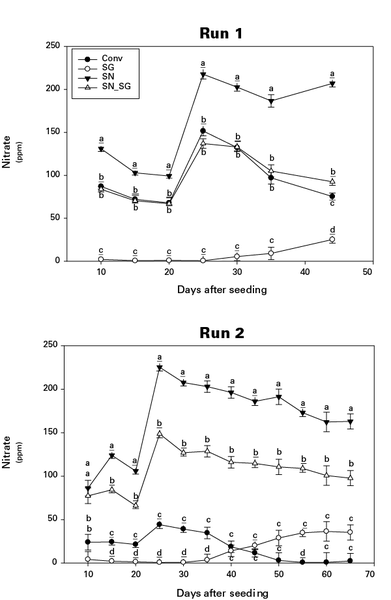
Ammonium-N float water concentration was greatest in SG treatments 25 days after seeding but declined rapidly over the following 20 to 30 days (Figure 1). In Figure 1, the means with the same letter within the same run and day after seeding are not significantly different (α = 0.05). Fertility treatments include a conventional control (Conv), Peruvian SG + K sulfate (SG), sodium nitrate + Peruvian SG + K sulfate (SN SG), and sodium nitrate + K sulfate (SN). The decline in ammonium concentration was complemented by an increase in nitrate-N concentration during the same period (Figure 2), indicating ammonium conversion to nitrate. In Figure 2, the means with the same letter within the same run and day after seeding are not significantly different (α = 0.05). Fertility treatments include a conventional control (Conv), Peruvian SG + K sulfate (SG), sodium nitrate + Peruvian SG + K sulfate (SN_SG), and sodium nitrate + K sulfate (SN).
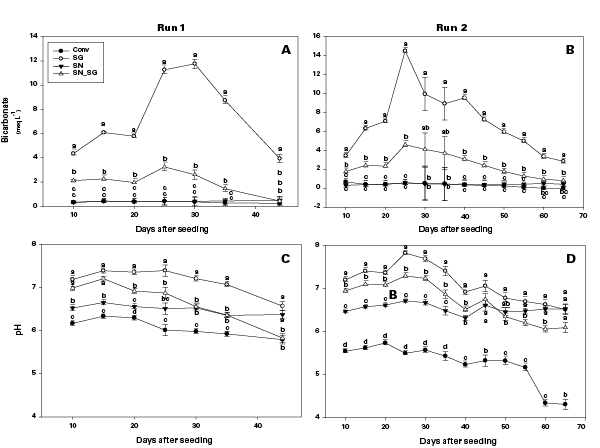
Bicarbonate concentration was greatest in SG only (≥12.0 meq L-1) and SG+SN (≥3.0 meq L-1) treatments 25 days after seeding, but was <1.0 meq L-1 in SN-only treatments, further implicating SG as a source of bicarbonates in organic float systems (Figure 3). In Figure 3, means with the same letter within the same run and day after seeding are not significantly different (α = 0.05). Fertility treatments include a conventional control (Conv), Peruvian SG + K sulfate (SG), sodium nitrate + Peruvian SG + K sulfate (SN SG), and sodium nitrate + K sulfate (SN). The established bicarbonate limit is 2.0 meq L-1, beyond which acidification is recommended. Despite the high bicarbonate concentrations documented in SG treatments, seedling growth was not impacted. Ultimately, SG- and SG+SN-based fertility programs produced seedlings comparable to 16-5-16 and appear to be suitable for the production of organic tobacco seedlings (Table 3).
|
Treatmenta |
Usable Plantsb (%) |
Stem Diameter (mm) |
Stem Height (cm) |
|
Stagnant |
86.7 |
2.8 abc |
5.97 a |
|
Aerator |
87.2 |
2.9 ab |
5.62 ab |
|
Sulfuric acid |
91.5 |
2.9 a |
5.56 abc |
|
Acetic acid |
91.1 |
2.7 b |
4.58 bc |
|
Liquid citric acid |
87.8 |
2.8 ab |
4.53 c |
|
Granular citric acid |
87.7 |
2.8 ab |
5.35 abc |
|
a Acidifier treatments were applied every 10 days throughout the duration of the study and rates determined based on float bed solution pH. |
|||
These fertility programs should be managed to include additional nutrients, such as P, to provide a complete nutrition program. Fertility programs lacking appropriate P will result in poor growth and unusable plants regardless of N availability (Figure 4). Furthermore, bicarbonates should be monitored and corrected accordingly. While they did not appear to cause any negative effects, dissolved oxygen values in the SG and SG+SN treatments dropped precipitously after the final fertilizer application. This drop is most likely caused by high rates of respiration in the microbial populations in the float system solution.
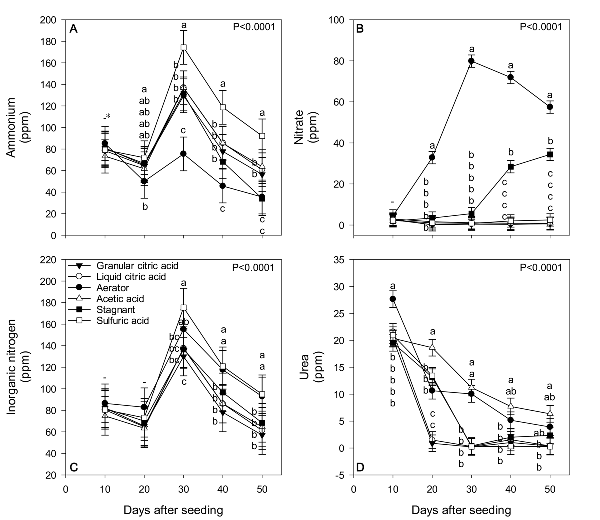
Separate, complementary float system studies were conducted to address the low dissolved oxygen and high bicarbonate concentrations associated with organic fertilizer mineralization. Experimental design and layout were identical to the prior described study, however, only the PG+PS fertilizer treatment was used, as this treatment led to the highest bicarbonate concentrations (Figure 3). Three OMRI-listed acidifying agents were investigated: liquid citric acid (BioLink® Acidifier, 50% a.i.; Westbridge Agricultural Products, Vista, California), granular citric acid (food grade citric acid, 99.5% a.i.; LD Carlson Company, Kent, Ohio), and acetic acid (Green Gobbler, 30% a.i.; Green Gobbler, Copiague, New York). Sulfuric acid (battery fluid acid, 36.5% a.i.; East Penn Manufacturing, Lyon Station, Pennsylvania) was used as a conventional check, as this product is commonly used as an acidifying agent in conventional tobacco float beds. Finally, stagnant (no acidifier, no movement) and aerated treatment containing a constantly running aeration device (H2ShOw bubble maker; Hydor Group, Sacramento, California) were used as controls, giving a total of six treatments. Application rates of 5.5 ml ml-1 HCO3- L-1 (sulfuric acid), 43 ml ml-1 HCO3- L-1 (acetic acid), 7.5 ml ml-1 HCO3- L-1 (liquid citric acid), and 4.5 g ml-1 HCO3- L-1 (granular citric acid) were predetermined by North Carolina Department of Agriculture & Consumer Services lab tests.
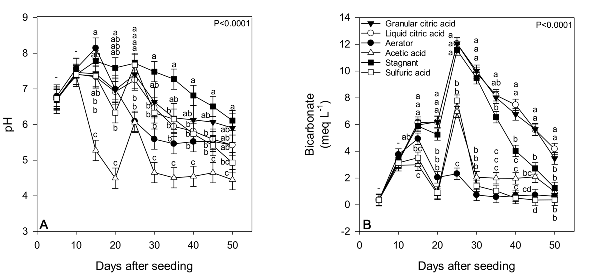
Acidifier treatments significantly affected solution pH and bicarbonate concentrations. Generally speaking, acetic acid led to consistently lower pH throughout the study (Figure 5). The simple addition of aeration led to the lowest bicarbonate concentrations throughout the study, which were below the 4 meq L-1 threshold. Both liquid and granular citric acid along with the stagnant control led to high bicarbonate concentrations. In Figure 6, means with the same letter within the same day after seeding are not significantly different (Tukey’s HSD; α = 0.05). Sulfuric acid, acetic acid, liquid citric acid, and granular citric acid treatments were applied every five days at rates calculated to reduce solution bicarbonate levels below 2 meq L-1. Dashes denote no significant differences among means within a specific time.
The addition of aeration created an environment that favored mineralization and nitrification, resulting in the production of higher nitrate-N as compared to other treatments (Figure 6). Conversely, addition of sulfuric acid led to significantly higher concentrations of ammonium and negligent nitrate throughout the entirety of the study. The conversion of organic N source material to nitrate is facilitated in part by nitrifying bacteria, after first being released as ammonium through decomposition of the organic source. These organisms take ammonium or nitrite and convert it to nitrate, through an oxygen-dependent process. Because these bacteria require oxygen, the maintenance of high dissolved oxygen concentrations via the aerator most likely created a float bed solution environment that favored mineralization and nitrification of organic fertilizer source material. In Figure 6, means with the same letter within the same day after seeding are not significantly different (Tukey’s HSD; α = 0.05). Sulfuric acid, acetic acid, liquid citric acid, and granular citric acid treatments were applied every five days at rates calculated to reduce solution bicarbonate levels below 2 meq L-1. Dashes denote no significant differences among means within a specific time.
Even with high differences in bicarbonate and nitrate concentrations, we did not observe appreciable differences in transplant usability (Table 3). Starting water quality, in terms of pH and bicarbonate concentrations, was acceptable; baseline pH and bicarbonate concentrations were 5.97 and 0.32 meq L-1. The elevated bicarbonate concentrations observed in this study were a result of fertilizer interaction and degradation and not an inherent water quality issue (Figure 6). For float systems, farmers may use source water with naturally occurring elevated bicarbonate concentrations. We did not investigate how well aeration can address inherently high bicarbonate concentrations and thus do not know how well aeration would work under this circumstance.
Additional Points for Consideration
- Producers might consider processing SG prior to application. Smaller SG particles will have a higher surface area and will be more water-soluble, both of which should increase N release.
- When blending N sources, target 40 ppm P from SG (4.85 oz. 12-11-2/100 gallon float water). This will ensure adequate P for the seedlings when added to the float water in two separate applications.
- Consult with your local Extension agent if you suspect a deficiency (such as calcium or boron). Organically approved secondary and micronutrient sources are available but need to be confirmed by an organic certifying entity.
- Consult with your organic certifier prior to the use of ANY fertilizer source.
- Water circulation is critical for organic nutrient sources, as some (guano) are not easily dissolved. Submersible pumps will help circulate water and nutrients and can add oxygen to the float water. The addition of oxygen is recommended, as it will help promote mineralization and nitrification and can potentially reduce bicarbonate concentrations.
- Float water samples should be collected and analyzed at frequent intervals.
Publication date: March 8, 2021
AG-892
Recommendations for the use of agricultural chemicals are included in this publication as a convenience to the reader. The use of brand names and any mention or listing of commercial products or services in this publication does not imply endorsement by NC State University or N.C. A&T State University nor discrimination against similar products or services not mentioned. Individuals who use agricultural chemicals are responsible for ensuring that the intended use complies with current regulations and conforms to the product label. Be sure to obtain current information about usage regulations and examine a current product label before applying any chemical. For assistance, contact your local N.C. Cooperative Extension county center.
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.