Background and Description
European red mite (Panonychus ulmi; ERM) is found on fruit trees, shade trees, and shrubs worldwide. It is the most common mite occurring on apple trees in the Southeast.
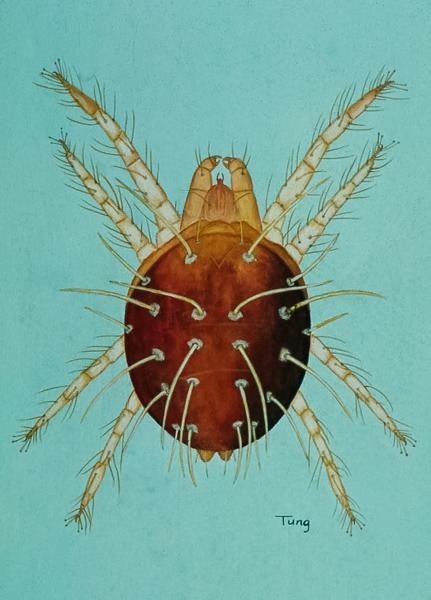
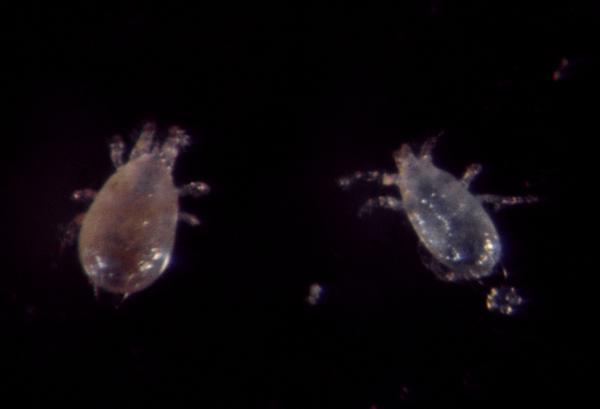
Adult female ERM have elliptical bodies that are approximately 1/64-inch long (0.4mm, not much larger than a period) and range from bright to brownish red, with four rows of curved spines on their backs. Males are slightly smaller, more lightly colored, and have abdomens that are more pointed than curved. Both female and male adults have eight legs, but freshly hatched nymphs have only six legs. Later nymphal stages have eight legs. In the summer, eggs are laid on leaves and appear as single, translucent spheres with distinct white stalks that are visible only under a microscope. Overwintering eggs are bright red and are laid in crevices of twigs or bark. Eggs are extremely small, about 1/200 inch (0.13mm) in diameter.
Life history
Overwintering ERM eggs begin hatching right before bloom. Mites then move onto leaves and feed on the tissues. Females lay roughly one egg per day - if a female has mated, the fertilized eggs develop into both male and female mites; if she has not mated, the unfertilized eggs develop into males. Eggs hatch into six-legged larvae, then progress through eight-legged protonymph and deutonymph stages before becoming eight-legged adults. Usually ERM populations build up slowly during the spring, increasing to unacceptably high levels only if summer conditions are favorably hot and dry. Eight to ten generations may develop per year.
Damage
ERM feed on leaves. When populations are severe, leaves may lose color or become brown (a condition referred to as "bronzing"). This can lead to fewer fruit, dropped fruit, and lower fruit quality, as well as a lower return bloom the following season.
Many factors determine the severity of a mite infestation, including the time of year when injury occurs, the duration of feeding, the trees' vigor and cultivar, crop load, and weather conditions. The earlier that foliage is injured (i.e., May or early June), the more detrimental the damage will be to tree health. Midseason (i.e., July or later) injury is less significant, but can combine with other stresses to cause fruit drop, poor fruit color, or reduced effectiveness of growth regulating chemicals.
Monitoring and Control
Bio-control: Several beneficial arthropods can help keep ERM and twospotted spider mite populations below damaging levels. The most common in the Southeast are a phytoseiid mite (Neoseiulus fallacis) and the complex of generalist predators (e.g., black lady beetles (Stethorus punctum) and lacewings). However, recent research in North Carolina suggests that neither of these predators overwinters to any significant degree within orchards, so they must be reestablished in orchards in the spring. Hence, practices that delay the buildup of ERM and enable predators to increase before mites become a problem will favor biological control. The two most effective practices are applying a delayed dormant oil spray and avoiding insecticides toxic to these predators.
Monitoring Mite Populations: Use a regular monitoring program to follow the buildup of pest mite populations and to determine if and when supplemental applications of a miticide are necessary to avoid economic damage. Monitor each contiguous block of apples weekly beginning when adult mites first appear (which may vary from mid May to early July). Within each block, examine 5 leaves from each of 10 trees with a visor lens or hand lens. Rather than counting the total number of mites on each leaf, record the number of leaves infested with one or more mites, and estimate the mite density on a per-leaf basis from the table below.
| % Mite-infested leaves (1+ mite/leaf) | Expected number of mites per leaf |
|---|---|
| 40 | 0.7 |
| 45 | 0.9 |
| 50 | 1.1 |
| 55 | 1.3 |
| 60 | 1.5 |
| 65 | 2.0 |
| 70 | 2.6 |
| 75 | 3.4 |
| 80 | 4.7 |
| 85 | 6.8 |
| 90 | 11.4 |
| 95 | 26.4 |
Determining the Need for Miticides: When mite populations reach a density of 5 to 10 mites per leaf (80 to 90 percent infested leaves) decide whether to use biological control or a miticide to prevent mites from increasing to higher densities. Count the actual number of N. fallacis on sample leaves with a visor lens. If the ratio of N. fallacis to ERM is between 1 to 5 and 1 to 15, biological control is possible. For biological control with S. punctum to occur, the ratio should be 2.5 S. punctum to 1 ERM. S. punctum should be sampled by counting the number of adults and larvae observed during a timed 3-minute search around the periphery of mite-infested trees. S. punctum larvae must almost always be present if this predator is to control mites. If neither predator is present at sufficient levels for biological control to occur, and mite populations are between 5 to 10 mites per leaf, apply a miticide. In areas where Alternaria blotch is a problem on Delicious apples, biological control is usually not an option. In the presence of Alternaria blotch, mite populations must be maintained at very low levels to avoid high levels of Alternaria and premature defoliation. If preventive control measures are not used, a modified threshold level of 1 to 2 mites per leaf should dictate the need for miticides.
See the "Pest and Orchard Management Program" and "IPM Practices for Selected Pests" section of the Integrated Orchard Management Guide for Commercial Apples in the Southeast for the most current control guidelines.
Publication date: Feb. 23, 2015
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.