Introduction
Angular leaf spot disease of strawberry, caused by the bacterium Xanthomonas fragariae, is often confused with common leaf spot and leaf scorch diseases. Once infection is established, little can be done until the wet cool conditions subside. Fortunately, this disease generally does not affect yields severely.
Symptoms and Signs
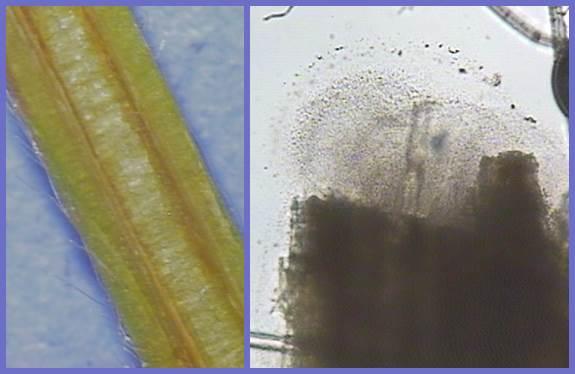
Water-soaked lesions first appear on the lower surface of the leaf, becoming angular as they enlarge and usually delineated by veins. When conditions are very moist, lesions may exude a viscous yellow substance that is actually a mass of bacteria. Upon drying, a characteristic white film is left on the leaf surface. In time, lesions will also be visible on the upper leaf surface as irregular, reddish brown spots that may be surrounded by a yellow halo. These symptoms are difficult to distinguish from common leaf spot and leaf scorch. One identifying characteristic is the translucent nature of lesions when leaves are held up to a bright light; looking from the backside, light will pass through the angular lesions. Entire leaves and major veins may become infected, giving the leaves a ragged appearance and plants can be stunted (Figure SS-2). Berry caps may become infected, darkened, and unappealing (Figure SS-3). Vascular infection (Figure SS-4) and wilting by X. fragariae may lead to plant death, but this is not common. Infected tissue can be finely chopped and viewed under a microscope; lesions will ooze masses of bacteria (Figure SS-4).
Disease Cycle
X. fragariae primarily enters the field via infected planting stock, and may persist in the field by overwintering or oversummering in infected plants and dead leaves. In the Southeast, problems have not persisted from one year to the next in annual plantings due to soil inoculum. The pathogen cannot survive freely in the soil, but can survive on transplants in cold storage for one year and on plant debris through long dry periods. Bacteria become active and are splash-dispersed to healthy leaves in wet weather or with irrigation water. X. fragariae favors low day (60°F) and night temperatures (near freezing) and high relative humidity. Favorable conditions for disease development occur during transplant establishment and when frost protecting. Healthy plant tissues are more likely to become diseased than stressed tissues. In cool wet springs the pathogen builds up on leaf tissue and splash disperses to the strawberry calyx (cap) where angular lesions can occur. Symptoms decline as temperatures increase.
Management
Cultural
1. Site Selection
Choose a site with good air circulation and sun exposure to promote drying of foliage.
2. Use Disease-Free Plants
Use certified plant material or plant material grown under similar strict conditions. Be aware that infected transplants may not exhibit signs of infection until exposed to a more favorable climate, such as exists in the southern states. Tests are currently being developed to detect infected nursery stock. Resistance to angular leaf spot exists in some genotypes, yet no commercially desirable cultivars contain high levels of resistance.
3. Monitor and Husbandry
Control weeds to allow air to circulate freely around plants. In fields with known infections, reduce overhead irrigation events for frost protection and rely on floating row covers more. Avoid using overhead irrigation if possible. Under serious disease conditions, ensure all strawberry debris is soil incorporated to optimize tissue break down. Rotation is not essential.
Chemical Control
Application of registered copper materials prior to rapid growth may reduce disease, but such products are not very effective in managing angular leaf spot because the bacteria can reside out of reach within the plant tissue. During cool wet springs, three applications during early fruit development may suppress lesion development on the caps. Caution should be taken when using copper fungicides because accumulations can be phytotoxic. Actigard is also registered and induces plant defences to limit bacterial colonization in the plant. For specific products and rates of use see our Strawberry IPM Guide for current recommendations.
Pathogen
Xanthomonas fragariae is a gram negative, aerobic, rod-shaped bacteria with cells measuring 0.4-1.0 µm wide by 0.7-2.0 µm long. Colonies of X. fragariae usually are mucoid, convex, and yellow when grown on yeast dextrose calcium carbonate (YDC) media or sucrose peptone agar (SPA) (Maas, 1998). All members of the genus Xanthomonas are catalase positive and oxidase negative (Schaad et al., 2001).
Diagnostic Procedures
To avoid confusing symptoms of abiotic injury or fungal infection to those of bacterial leaf spot, the shape of the lesion is important to note. The lesion shape of angular leaf spot is defined by leaf veins and veinlets, so during fairly early stages the shape is angular rather than the round shape typical of fungal leaf spots or injury caused by some pesticides or other chemical sprays. Another important distinguishing characteristic of Angular leaf spot is that the lesions are translucent when viewed with transmitted light but dark green and water-soaked when viewed with reflected light.
Bacterial streaming can be observed from infected leaf tissue by making a cross-sectional cut through a leaf lesion, placing the tissue in a droplet of water, placing a cover-slip over the sample, and examining it under a bright- or dark-field microscope (~100x). This can also be done with vascular tissue. Streak-plate isolations can be made from expanding lesions or vascular tissue on sucrose peptone agar (SPA) and yeast dextrose calcium carbonate agar (YDC). Allow the bacteria to grow for at least 48 hours at room temperature and examine periodically for colony growth before subculturing. Colony color and appearance, Gram reaction (using a 3% KOH solution) and the oxidase reaction are easy tests for preliminary confirmation when a Xanthomonas species is suspected.
ELISA tests are available from AC Diagnostics for X. fragariae. Agdia also offers a generic ELISA test for Xanthomonas. ELISA testing can be performed on bacterial cultures or directly on plant tissue.
Single gene sequencing (e.g. partial sequencing of the Gyrase B and other genes), once fully validated, is likely to offer rapid and accurate identification of all Xanthomonas species and pathovars.
References
Maas, J. L. 1998. Compendium of Strawberry Diseases, 2nd edition. APS Press. St. Paul, MN.
Schaad, N. W., Jones, J. B., and Chun, W. 2001. Laboratory guide for identification of plant pathogenic bacteria. APS Press St. Paul, MN.
Publication date: July 24, 2014
Reviewed/Revised: July 26, 2024
There is an alternate Spanish language version of this document here: Mancha angular de la fresa
N.C. Cooperative Extension prohibits discrimination and harassment regardless of age, color, disability, family and marital status, gender identity, national origin, political beliefs, race, religion, sex (including pregnancy), sexual orientation and veteran status.